Application field
Production of acetonitrile by ammoniation of acetic acid
Background technique
The most important use of acetonitrile is as a solvent, such as extracting butadiene, isoprene extractant, spinning solvent for synthetic fibers and solvents for certain oils, phenols and colored substances; Solvent for extracting fat from animal and vegetable oils; used as reaction medium for recrystallization of steroid drugs in medicine, etc. Acetonitrile is also an intermediate of medicine (vitamin B1) and spices, and a raw material for preparing 2-picoline, triazine, ethylamines, dipropionitrile, imidazole, and propylene bisacetonitrile.
In the traditional process of synthesizing refined acetonitrile by ammoniation of acetic acid, the acetic acid and ammonia gas are first vaporized and then put into the reaction kettle, and the ammoniation reaction takes place under the action of a catalyst with aluminum oxide as the carrier to obtain acetonitrile. After the reaction, the crude product contains acetonitrile, water and a small amount of ammonia gas. After the ammonia gas is absorbed and removed by the two-stage absorption tower, it enters the concentration tower, negative pressure tower and positive pressure tower to obtain the finished acetonitrile.
Since acetonitrile will form azeotrope with water (the water content of acetonitrile-water azeotrope under normal pressure is about 16 wt.%), the acetonitrile mother liquor in the absorption section cannot be directly refined into finished acetonitrile by conventional distillation methods (refined acetonitrile Moisture content is usually ≤0.1 wt.%). At present, the methods for refining acetonitrile in the ammonification of acetic acid to acetonitrile mostly use a concentration tower, a negative pressure tower and a positive pressure tower. This type of process consumes a lot of steam, occupies a large area of equipment, has a long process flow, a large amount of material and liquid circulation, complicated operation, and low acetonitrile yield.
Traditional crafts
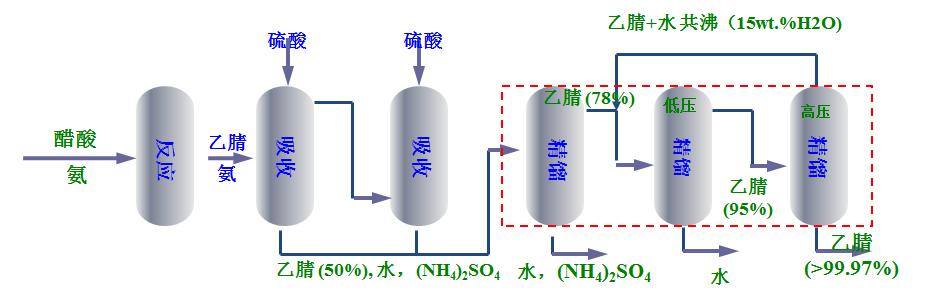
New Technology
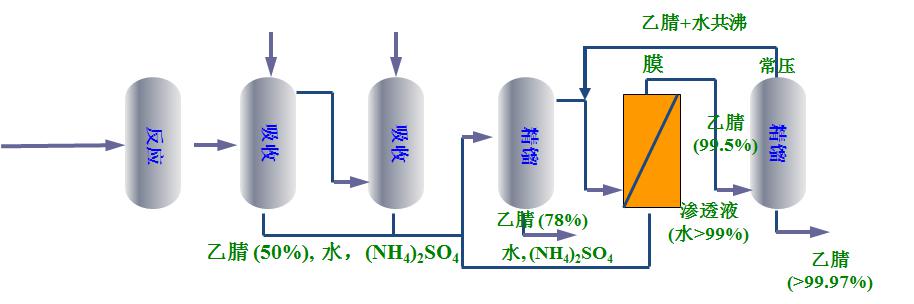
Technical advantages
The new process developed by Jiutian Hi-Tech has high recovery rate, simple process, high safety factor, high energy utilization rate, small circulation of material and liquid, and low energy consumption in operation (traditional process steam consumption 4t/t, new process 1.6~2t/t ), without the addition of the third component.
Project case
Shandong Huihai, Nantong Liyang

Shandong Huihai 10,000 tons/year acetonitrile plant
Hydrogenation of acetone to produce isopropanol
Background technique
Isopropanol is an important chemical product and raw material with a wide range of uses. China is the largest importer of isopropanol in Asia. At present, the annual output of isopropanol in my country is about 160,000 tons. As the production of isopropanol is restricted by many factors such as raw material supply, production technology, and environmental protection, the domestic annual output is far less than annual market demand. In 2010, the import volume of isopropanol was 115,000 tons, and in 2011, the import volume of isopropanol was 100,000 tons. The direct hydration of propylene is the main method for industrial production of isopropanol. It is to hydrate propylene directly in the presence of a catalyst to generate isopropanol, while producing n-propanol by-product. Due to the shortage of domestic propylene resources, the consumption of propylene and energy consumption in the production of isopropanol by the propylene hydration process are very high, and the production cost is very high. Contrary to the situation of propylene, the domestic production capacity of acetone is large, the market volume is abundant, and the price is relatively low. Almost all acetone in industry is obtained by peroxidation of cumene (co-production with phenol). Due to the increase in the demand for phenol, a large amount of acetone is co-produced, and there is an imbalance in demand, which often leads to an excess of acetone, making the price of acetone lower than the price of isopropanol and propylene. Development of acetone hydrogenation to produce isopropanol The technology has broad application prospects.
During the hydrogenation of acetone to produce isopropanol, there are a small amount of side reactions. The side reaction products mainly include water, diisopropyl ether, diacetone alcohol, and 4-methylpentanol. These by-products are separated. The refining of isopropanol generally adopts two rectification towers for separation operation; the by-product water and a small amount of unreacted acetone are removed from the top of the first tower, and isopropanol and other heavy component by-products are obtained from the bottom of the tower. The isopropanol product with a water content of <0.03wt.% is obtained at the top of the tower, and the rest of the heavy components are removed in the bottom of the tower. Since isopropanol forms a ternary azeotrope with water and acetone (acetone 8wt.%, isopropanol 81wt.%, water 11wt.%), traditional azeotropic distillation, extractive distillation and other dehydration separation processes are complicated to operate and cost It is too high and the environmental pollution is serious, so that the current manufacturers generally sell it as waste liquid, causing the yield of isopropanol to decrease and the production cost to be too high.
Production Process

Typical engineering case

8,000 tons/year isopropanol/acetone/water film device for Yancheng Supur Chemical



